ESMO 2016 Poster Report of Apatinib Phase II Study Results
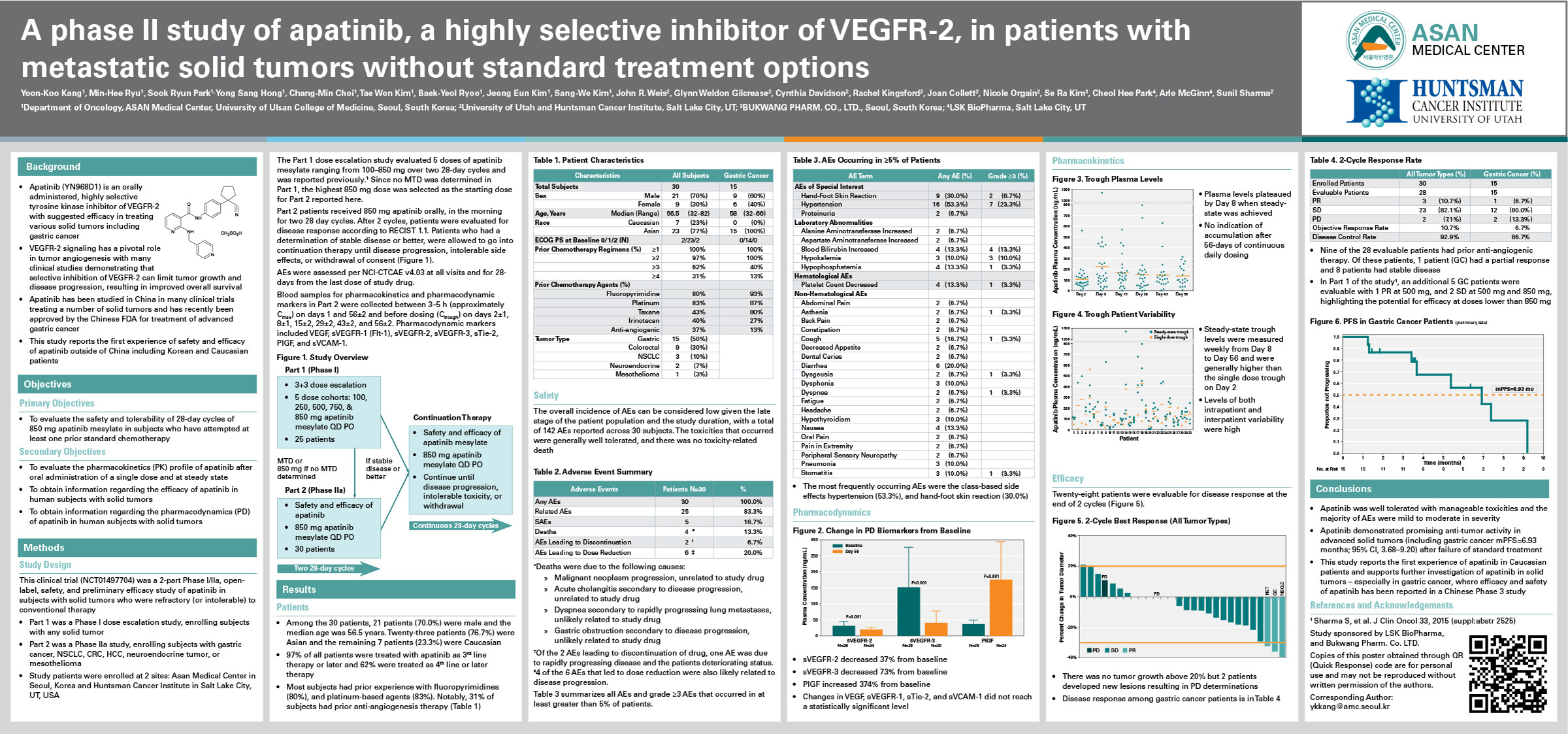
Apatinib (YN968D1) is an orally administered, highly selective tyrosine kinase inhibitor of VEGFR-2 in testing for the treatment of solid tumors including gastric cancer. Apatinib has been studied in China in many clinical trials treating a number of solid tumors and has recently been approved by the Chinese FDA for treatment of advanced gastric cancer.
This poster presented at ESMO 2016 reports the first experience of safety and efficacy of Apatinib outside of China including Korean and Caucasian patients. Overall, Apatinib was well tolerated with manageable toxicities and the majority of AEs were mild to moderate in severity. The preliminary efficacy in gastric cancer subjects indicated a mPFS of 6.93 months (95% CI, 3.68–9.20) after failure of standard treatment.
To read the full report, download the poster.